flowchart LR
subgraph IP["Infection Process"]
A[Residential segregation] --> B(Isolation)
A --> C(Concentration)
C --> D{Infection <br> Inequity}
B --> D
end
subgraph MP["Mortality Process"]
D --> E((Case Fatality <br> Rate))
E --> F{Mortality <br> Inequity}
B --> G(Healthcare <br> Access/Quality)
G .->
I(Comorbidity <br> Prevalence) .-> E
end
H{{Structural Racism}} --> A
H --> G
H --> I
style IP fill:none
style MP fill:none
Modeling to support health equity in Michigan
A systems of exposure approach
MICOM Site Visit
University of Michigan
2024-05-22
Jon Zelner
Dept. of Epidemiology
Center for Social Epidemiology and Population Health
University of Michigan School of Public Health
✉️ jzelner@umich.edu
🌐 epibayes.io


My goal today
Capitalize on having everyone together to pin down some ways that the health equity core can jumpstart an effective, accessible and sustainable analytic infrastructure that ensures equitable prevention and treatment in Michigan and beyond.
An unpleasant but urgent thought experiment
What if a new pandemic started today? 🐄
Do we have any expectation that the structural factors that led to enormous inequity in infection and death from SARS-CoV-2 are less potent now than 4+ years ago?
Are we prepared to prevent the worst inequities in infection, severe disease and death that characterized SARS-CoV-2 in Michigan?
Do we have a solid sense of what a pathogen with a different age-specific risk profile, i.e. a pandemic influenza variant more strongly impacting young children, will mean for inequity in infection and death?
Agenda
My argument for a ‘systems of exposure’ approach to modeling infection inequity in Michigan.
Applying this framework to SARS-CoV-2 data from Michigan.
Thinking ahead about what role the Health Equity Core should play in supporting the overall project.
Health Equity: A pillar? 🏗️ A cloud? ☁️ A web? 🕸️
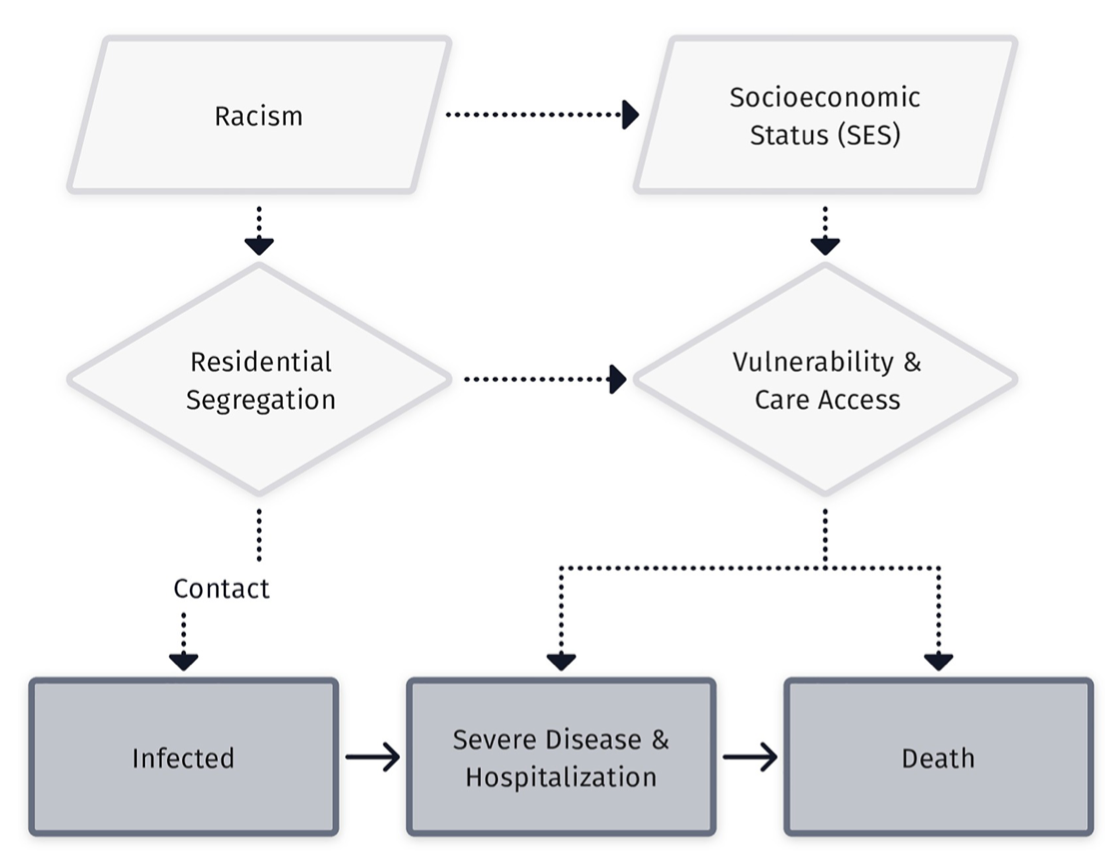
In the wake of the COVID-19 pandemic there has been a proliferation of conceptual frameworks linking socio-structural inequity to infection outcomes.
These are implicitly or explicitly rooted in the fundamental cause approach
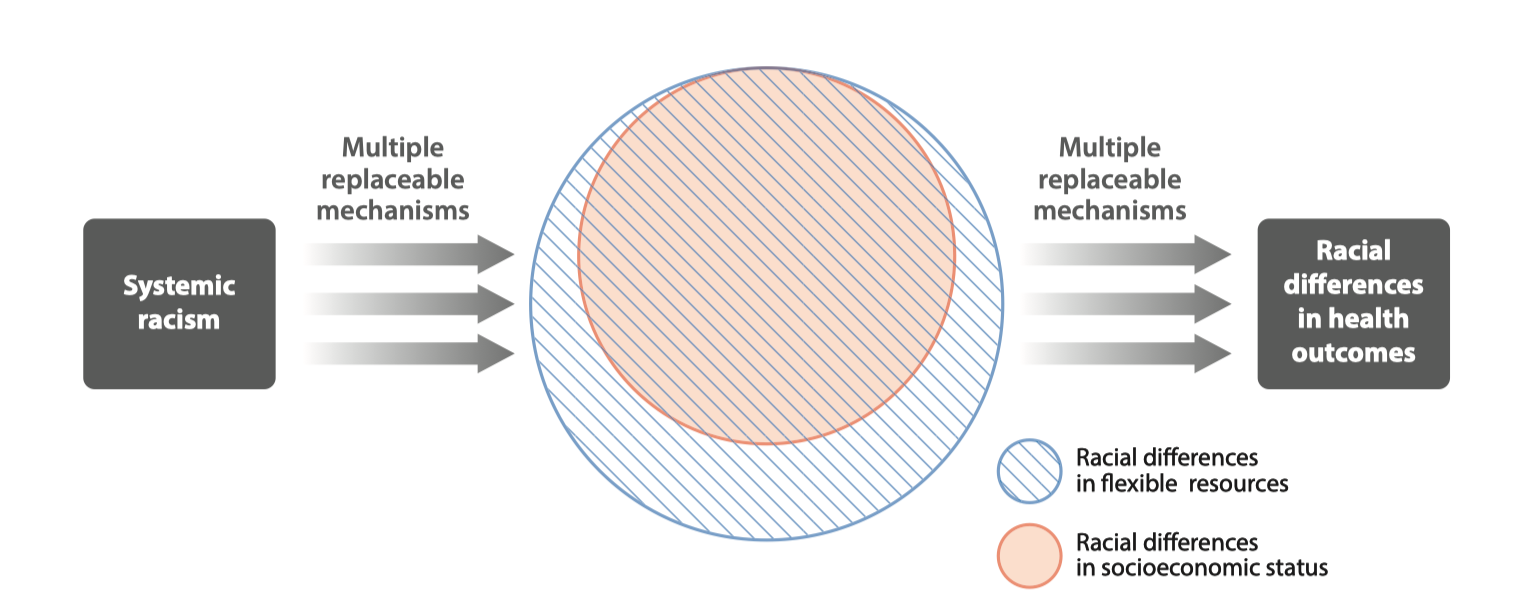
Conceptual model of racism as a fundamental cause of health inequity (From Phelan and Link (4))
What makes fundamental causes…fundamental?
Their effects persist even when intermediary mechansisms are addressed.
Simultaneously impact many intervening mechanisms and disease outcomes.
Innovations that are distributed unevenly increase inequity even if they reduce overall burden.
Wait, I thought innovation was a good thing?
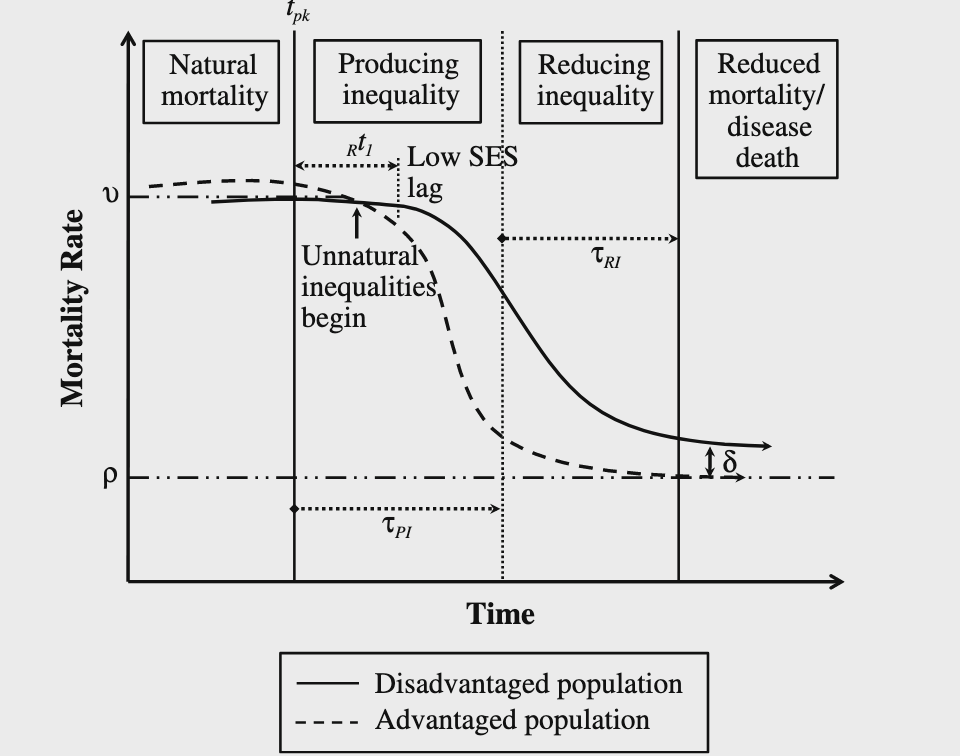
Innovations that diffuse along SES gradients may inadvertently increase inequity by providing early access to those who are most advantages (Figure from Clouston (2016) (5))
Fundamental causes orcheastrate infection inequity via systems of exposure
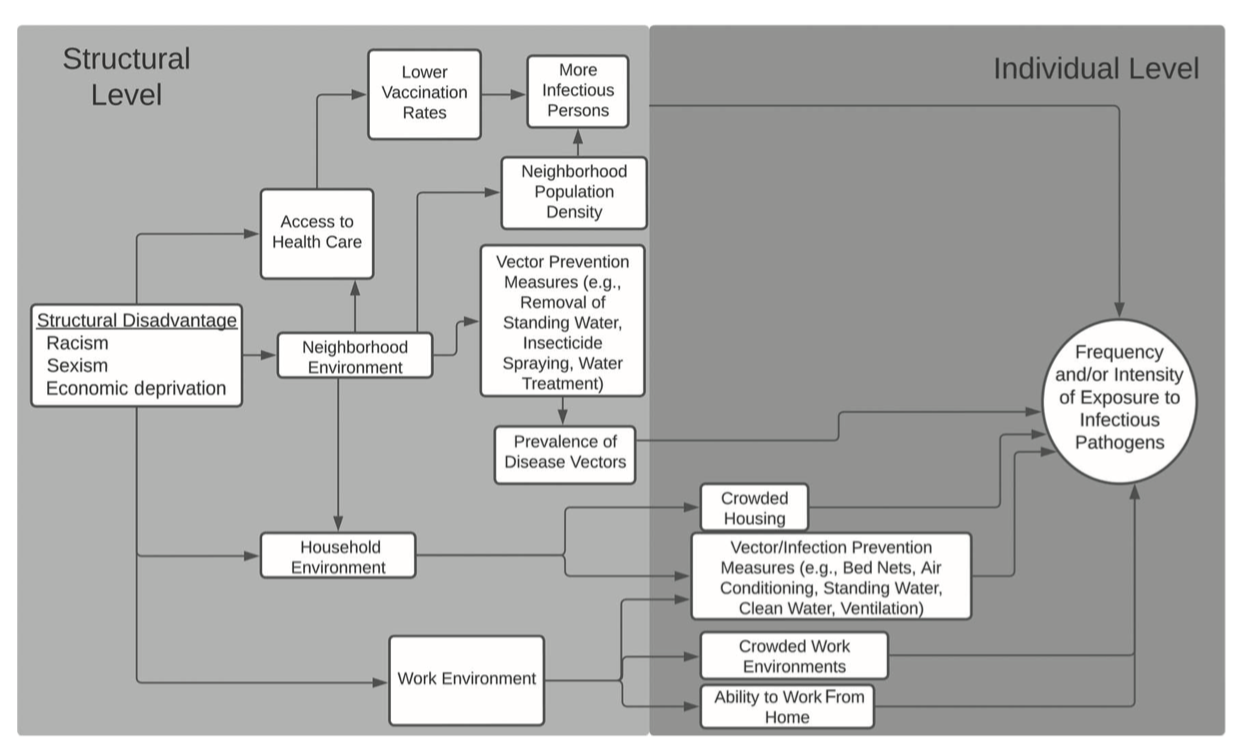
Riley (6) describes these systems in general terms relating to environmental exposure, social stress, differential treatment and so on.
In the context of infectious disease, this is about who gets exposed, when, and what happens to them when they are ill.
Requires a new approach to both transmission and statistical modeling (7).
What are the components of the systems of exposure that defined the COVID-19 pandemic?
Residential and occupational segregation that increased exposure.
Social and economic power to socially distance, work-from-home, etc.
Disparities in geographic access to healthcare.
Unequal treatment in healthcare settings.
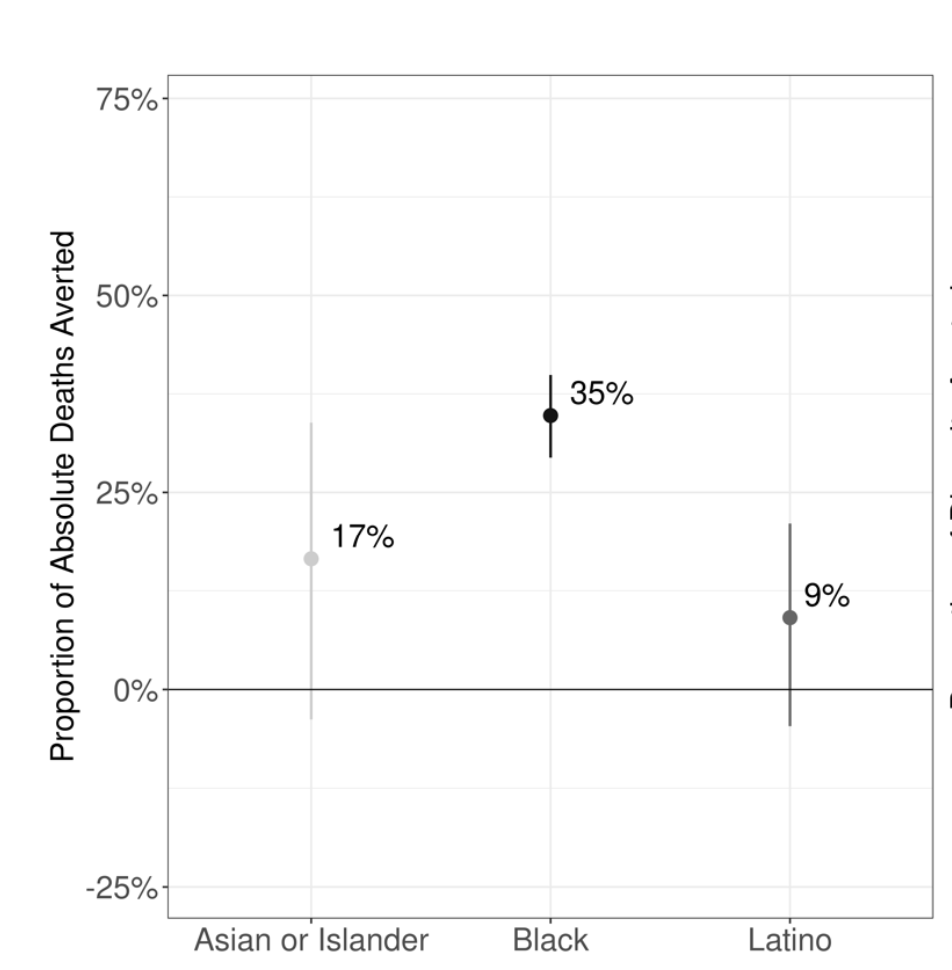
Case example: What explains time-varying patterns of SARS-CoV-2 mortality in Michigan during 2020?
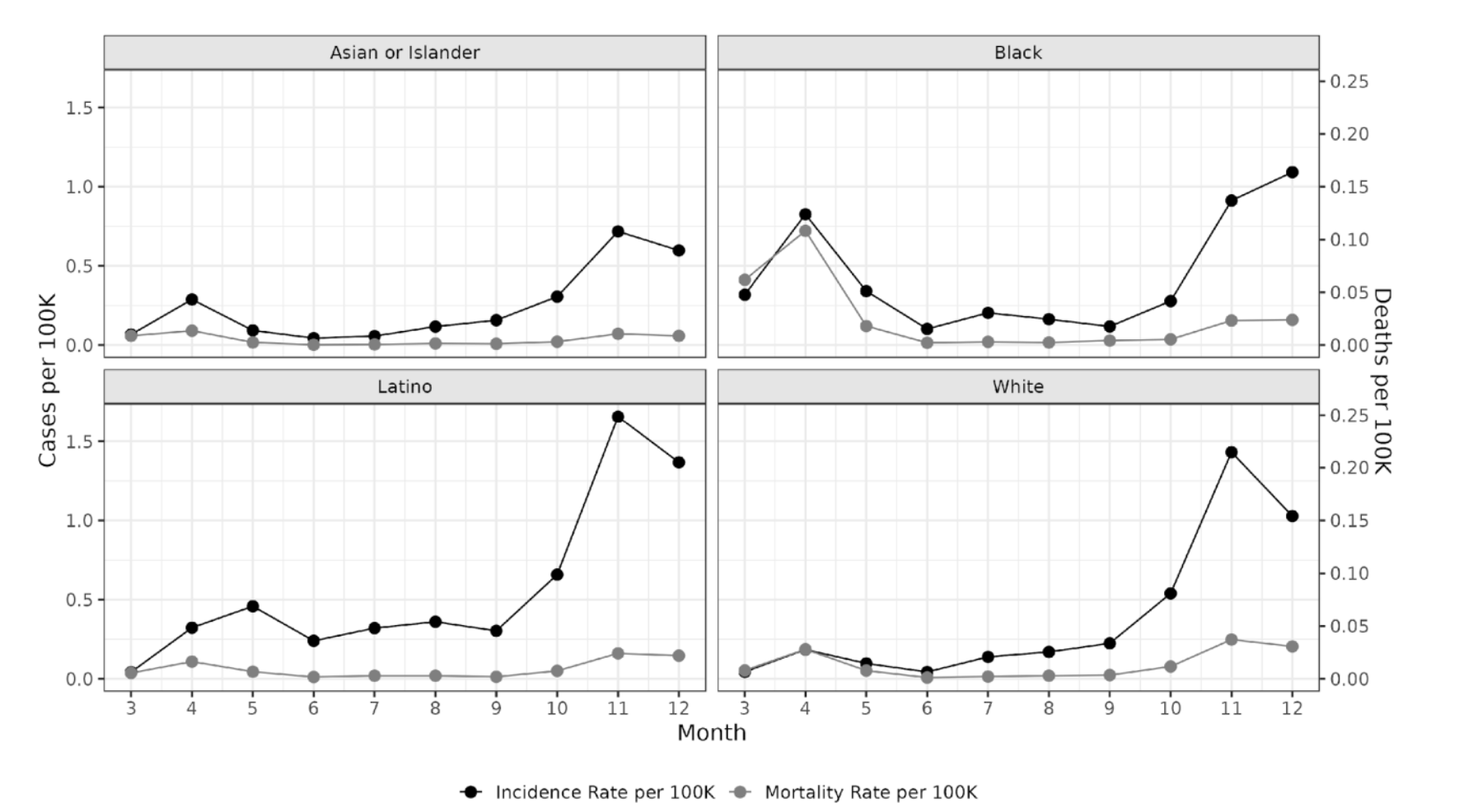
Differential incidence and mortality rates by race/ethnicity and month of 2020 (From Naraharisetti et al. Under Review.)
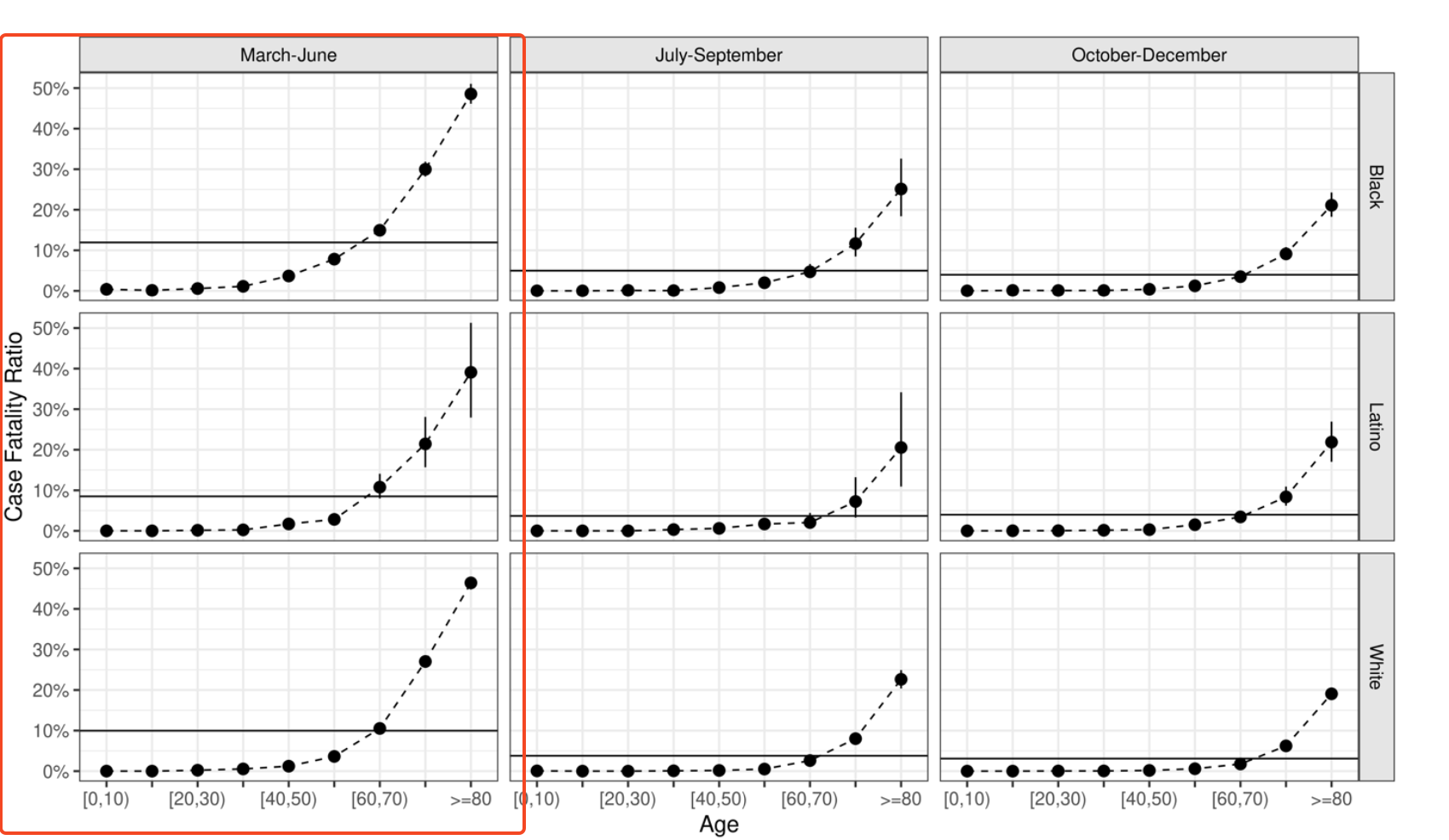
Case-fatality rates varied much more over time than they did between groups
Relative risks of infection were much greater in the first period than later periods
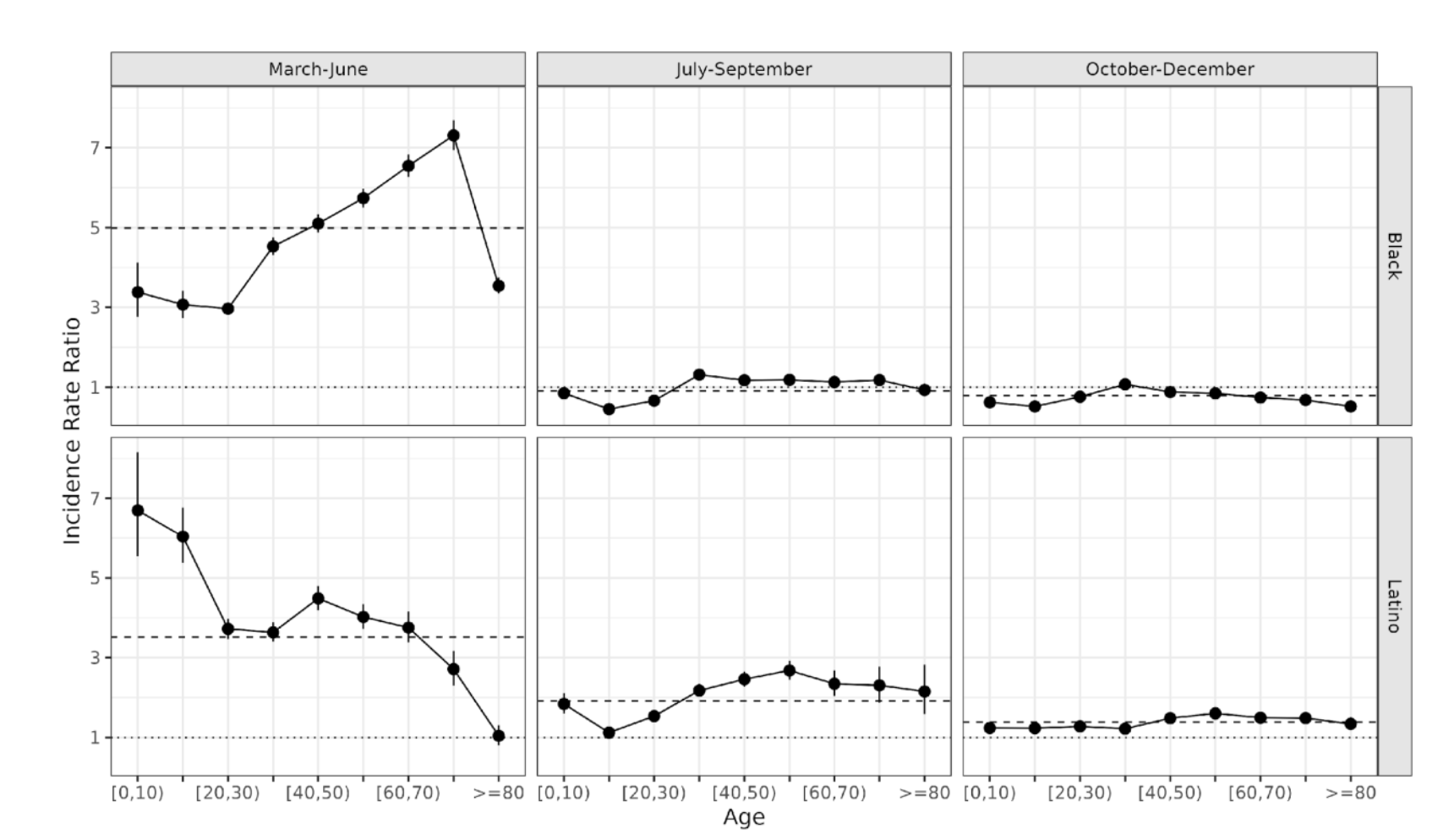
Infection timing matters
We can’t respond our way out of this type of outcome.
Our modeling efforts have to account for the structural preconditions to inequity in emerging infection risk.
But: We also have to be prepared for best- and wort-case scenarios in order to minimize and mitigate these inequities when they arise.
How should we be using models to think structurally about infection inequities?
Some implications of a systems of exposure approach
We need models that allow us to think about within- and between- epidemic comparisons.
We have to evaluate our efforts and results against an equitable outcome even if it hasn’t yet been realized.
Our models have to account for pathogen-specific interactions between the key social and biological dimensions of risk, e.g. age-specific risks of infection and death.
⚠️ Some of the challenges ahead ⚠️
Missing or absent data on individual socioeconomic status.
Incomplete administrative data (i.e. difficulty of obtaining detailed information on negative SARS-CoV-2 tests).
Increasing reliance on non-mechanistic metrics of social risks, e.g. social vulnerability index.*
Non-random missingness of key demographic covariates (e.g. race & ethnicity) (8)*.
Flaky population denominators that get worse as you break them down more.*